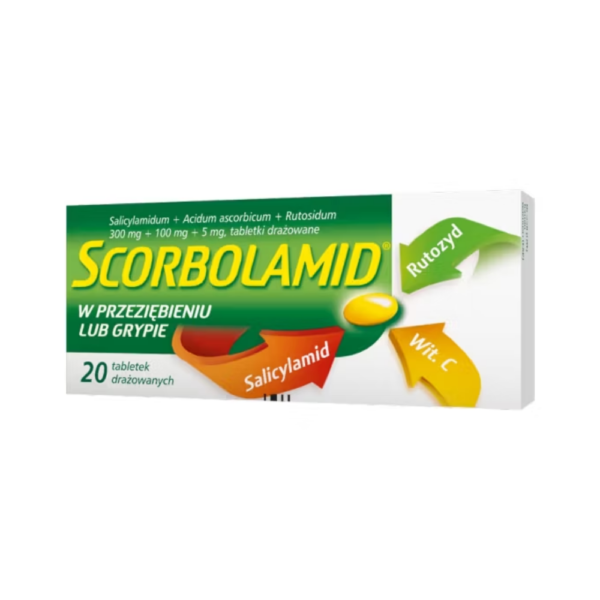
Scorbolamid
Scorbolamid is intended as a remedy for fever and pain associated with colds or flu, headaches, neuralgia.
Active substances: Acidum ascorbicum, Rutosidum, Salicylamidum
Composition of Scorbolamid
Each coated tablet contains: 300 mg of salicylamide ( Salicylamidum ), 100 mg of ascorbic acid ( Acidum ascorbicum ), 5 mg of rutoside ( Rutosidum ).
Excipients with known effect : sucrose and quinoline yellow (E104).
Action
Scorbolamide contains:
- Salicylamide – antipyretic, analgesic and anti-inflammatory.
- Rutoside – strengthens blood vessels and increases their elasticity. In addition, it protects vitamin C from oxidation and therefore vitamin C lasts longer.
- Ascorbic acid (vitamin C) – complements the deficiency of vitamin C, supporting the body’s immunity during infections.
Dosage
Adults and adolescents over 16 years of age:
Orally, usually 1 to 2 coated tablets 3 times a day.
The product is best taken with a meal or with a glass of water or milk.
The lowest effective dose should be used. Symptomatic treatment without medical advice should be carried out for the shortest possible time.
Contraindications of Scorbolamid
- Hypersensitivity to the active substances or to any of the excipients;
- Hypersensitivity to other non-steroidal anti-inflammatory drugs;
- Asthma with nasal polyps (caused or exacerbated by acetylsalicylic acid);
- Active gastric and duodenal ulcers, inflammations of the digestive tract;
- Acid-base balance disorders (diabetes, uremia, tetany);
- Blood clotting disorders (e.g. haemophilia, thrombocytopenia);
- Genetically determined deficiency of glucose-6-phosphate dehydrogenase (risk of haemolysis after long-term oral intake of ascorbic acid);
- History of kidney stones (risk of urinary stones after using high doses of ascorbic acid);
- Hemochromatosis, sideroblastic anemia, excessive amounts of uric acid in the blood or oxalate in the urine;
- Pregnancy;
- Breast-feeding.
The product should not be used in children under 16 years of age, except for rare diseases such as Kawasaki disease, due to the risk of Reye’s syndrome.
Contents
Round, biconvex, yellow coated tablets. The package contains 40 coated tablets.
Storage method
15°C – 25°C
Manufacturer
Warnings
This is medicine. For safety, use it according to the leaflet attached to the package. Do not exceed the maximum dose of the drug. If in doubt, consult your doctor or pharmacist.
The drug should be used with caution in bronchial asthma (possibility of aggravating asthma attacks), in liver or kidney failure and in patients with a history of peptic ulcer disease and gastrointestinal bleeding.
The product contains sucrose. Patients with rare hereditary problems of fructose intolerance, glucose-galactose malabsorption or sucrase-isomaltase insufficiency should not take this medicinal product.
Pregnancy
The safety of salicylamide in pregnant and breast-feeding women has not been established. However, as there is evidence of teratogenic effects of salicylamide in experimental animals even at low doses, the product should not be used in pregnant women.
Breast-feeding
The product should not be used during breastfeeding.
Driving
There are no data on contraindications to driving and using machines while using the product.
Overdosage
In animal studies, the toxicity of salicylamide and salicylates has been shown to be similar.
The lethal dose of salicylamide is 0.5-5 g/kg bw.
Central nervous system depression, hypotension, respiratory arrest, metabolic acidosis or hypoprothrombinemia may occur after an overdose of salicylamide.
In case of acute poisoning, the stomach should be emptied by vomiting or gastric lavage. Then administer activated charcoal and apply symptomatic treatment based on fluid replacement.
Interactions with other drugs
Salicylamide:
- Increases the concentration of acetylsalicylic acid and non-steroidal anti-inflammatory drugs (e.g. ibuprofen, diclofenac) in the blood serum;
- Intensifies side effects from the gastrointestinal tract during simultaneous use with corticosteroids;
- Intensifies the effect of oral antidiabetic drugs from the sulfonylurea group (tolbutamide, chlorpropamide), anticoagulants from the group of coumarin and heparin (increased risk of bleeding and prolonged bleeding time), methotrexate, valproic acid and sulfonamides;
- Reduces the effectiveness of drugs used in gout (probenecid, sulfinpyrazone).
Ascorbic acid:
- May reduce the effectiveness of warfarin and the plasma concentration of fluphenazine;
- Reduces the pH of the urine, which may affect the excretion of other, concomitantly used drugs.
Rutoside:
- Do not use with sulfonamides.
Side effects
The most common side effects, with the degree of severity depending on the dose of salicylamide, were observed from the gastrointestinal tract and the central nervous system. They occurred rarely at salicylamide doses of 325-600 mg, more often at higher doses.
Gastrointestinal disorders nausea, vomiting, abdominal cramps, burning sensation behind the sternum, anorexia, diarrhoea, irritation of the gastroduodenal mucosa (erosions and ulcers), dry mouth.
There was no clear association between the use of salicylamide and gastrointestinal bleeding.
Nervous system disorders Dizziness, drowsiness, headache.
Skin and subcutaneous tissue disorders facial flushing, rash, hyperhidrosis.
The product should be used with caution in patients who have experienced rash, angioedema, bronchospasm, rhinitis or shock with acetylsalicylic acid or other non-steroidal anti-inflammatory drugs. Although salicylamide has not been proven to cause all the side effects characteristic of salicylates, due to the similarity of structure and pharmacological action, there is a possibility of their occurrence.
Ascorbic acid reduces the pH of the urine, which facilitates the precipitation of urate, cystine or oxalate.

Reviews
There are no reviews yet.